 
: 275ff : 6 The defined precedent is for the lipophilic and hydrophilic phase types to always be in the numerator and denominator respectively for example, in a biphasic system of n- octanol (hereafter simply "octanol") and water: 
: 275ff When one of the solvents is water and the other is a non-polar solvent, then the log P value is a measure of lipophilicity or hydrophobicity. The partition coefficient, abbreviated P, is defined as a particular ratio of the concentrations of a solute between the two solvents (a biphase of liquid phases), specifically for un- ionized solutes, and the logarithm of the ratio is thus log P. Here, the green substance has a greater solubility in the lower layer than in the upper layer. For example, partition constant, defined asĪn equilibrium of dissolved substance distributed between a hydrophobic phase and a hydrophilic phase is established in special glassware such as this separatory funnel that allows shaking and sampling, from which the log P is determined. In contrast, the IUPAC recommends that the title term no longer be used, rather, that it be replaced with more specific terms. Nomenclature ĭespite formal recommendation to the contrary, the term partition coefficient remains the predominantly used term in the scientific literature. A related value, D, does not distinguish between different species, only indicating the concentration ratio of the substance between the two phases. If a substance is present as several chemical species in the partition system due to association or dissociation, each species is assigned its own K ow value. Partition coefficients can be measured experimentally in various ways (by shake-flask, HPLC, etc.) or estimated by calculation based on a variety of methods (fragment-based, atom-based, etc.). The partitioning of a substance into a solid results in a solid solution. Partition coefficients can also be defined when one of the phases is solid, for instance, when one phase is a molten metal and the second is a solid metal, or when both phases are solids. For example, the blood/gas partition coefficient of a general anesthetic measures how easily the anesthetic passes from gas to blood. If one of the solvents is a gas and the other a liquid, a gas/liquid partition coefficient can be determined. Conversely, hydrophilic drugs (low octanol/water partition coefficients) are found primarily in aqueous regions such as blood serum. Hydrophobic drugs with high octanol-water partition coefficients are mainly distributed to hydrophobic areas such as lipid bilayers of cells. Partition coefficients are useful in estimating the distribution of drugs within the body. Hence the partition coefficient measures how hydrophilic ("water-loving") or hydrophobic ("water-fearing") a chemical substance is. Most commonly, one of the solvents is water, while the second is hydrophobic, such as 1-octanol. In the chemical and pharmaceutical sciences, both phases usually are solvents. The partition coefficient generally refers to the concentration ratio of un-ionized species of compound, whereas the distribution coefficient refers to the concentration ratio of all species of the compound (ionized plus un-ionized). This ratio is therefore a comparison of the solubilities of the solute in these two liquids. In the physical sciences, a partition coefficient ( P) or distribution coefficient ( D) is the ratio of concentrations of a compound in a mixture of two immiscible solvents at equilibrium. There are many formulas to calculate the correlation coefficient (all yielding the same result).Not to be confused with distribution constant. You may use the linear regression calculator to visualize this relationship on a graph. Values close to -1 signal a strong negative relationship between the two variables. A value of 0 indicates that there is no relationship. 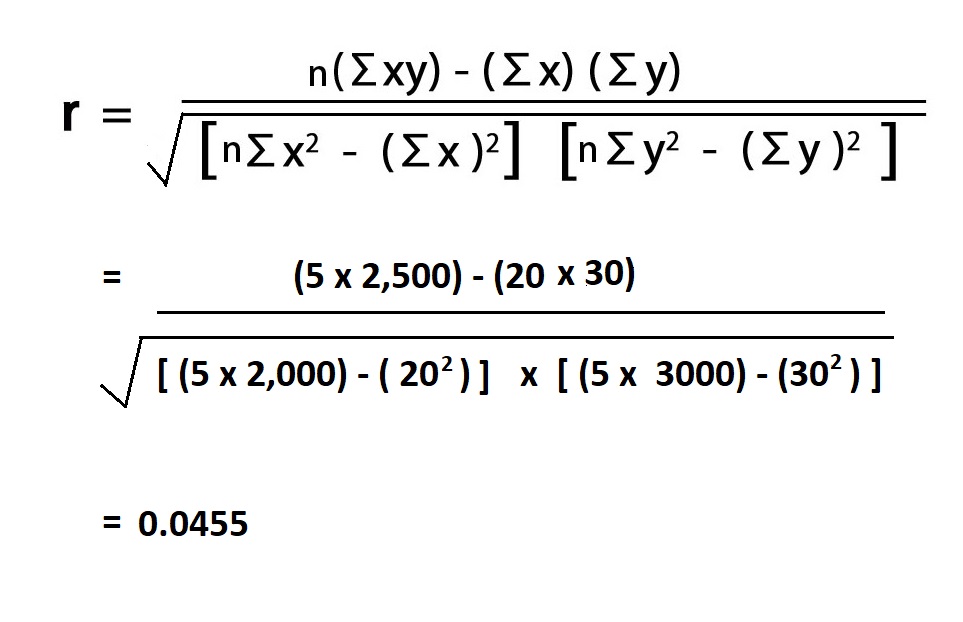
The correlation coefficient, or Pearson product-moment correlation coefficient (PMCC) is a numerical value between -1 and 1 that expresses the strength of the linear relationship between two variables.When r is closer to 1 it indicates a strong positive relationship. To clear the calculator and enter new data, press "Reset". The correlation coefficient will be displayed if the calculation is successful. Press the "Submit Data" button to perform the calculation. All x i values in the first line and all y i values in the second line:.You may enter data in one of the following two formats: This calculator can be used to calculate the sample correlation coefficient.Įnter the x,y values in the box above. Correlation Coefficient Calculator Instructions 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
